Obesity Pipeline, FDA Approvals, Clinical Trials Developments and Companies 2023 | Aardvark Therapeutics, Adocia, Agentix, AgeX Therapeutics, Altimmune, Amgen, Antag Therapeutics, and others.
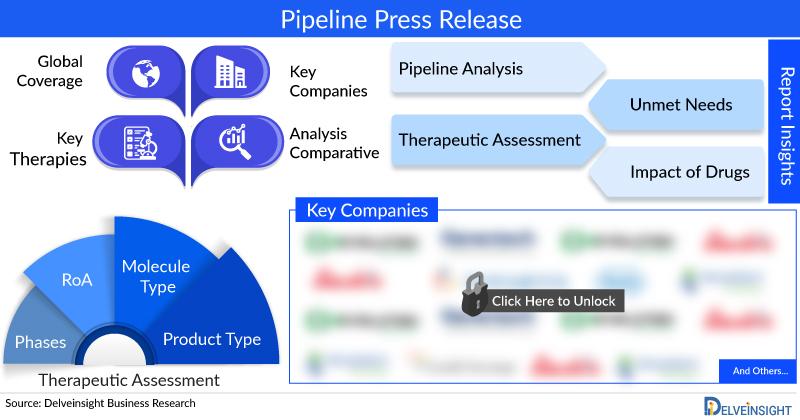
DelveInsight’s, “Obesity Pipeline Insight 2023” report provides comprehensive insights about 100+ companies and 130+ pipeline drugs in the Obesity pipeline landscape. It covers the Obesity pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Obesity pipeline therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive Obesity pipeline products in this space.
Key Takeaways from the Obesity Pipeline Report
- DelveInsight’s Obesity pipeline report depicts a robust space with 100+ active players working to develop 130+ pipeline therapies for Obesity treatment.
- The leading companies working in the Obesity Market include 180 Life Sciences, 9 meters, Aardvark Therapeutics, Adocia, Agentix, AgeX Therapeutics, Altimmune, Amgen, Antag Therapeutics, Aphaia Pharma, Aptorum Group, AstraZeneca, Bukwang Pharmaceutical, Caliway Biopharmaceutics Co Ltd, Can Fite Biopharma, Carmot Therapeutics, Inc., Cellivery Therapeutics Inc, CinFina Pharma, Clayton Biotech, Click Therapeutics, Corbus Pharma, DiscoveryBiomed Inc, Dong-A ST, Dongkook Pharmaceuticals, Eccogene, Elevian, Eli Lilly and Company, Empros Pharma, Enterin Inc., EraCal Therapeutics AG, ERX Pharmaceuticals, ERX Pharmaceuticals, Eternygen GmbH, Eurofarma, Gannex Pharma, Glaceum, Gmax Biopharm, GPER G-1 Development Group, LLC, Gubra Therapeutics, Hanmi Pharmaceutical, Hanmi Pharmaceuticals, Innovent Biologics, Jenrin Discovery, Jiangsu Hansoh Pharmaceutical, Kallyope, Kintai Therapeutics, Kriya Therapeutics, LG Life Sciences, Lipidio Pharmaceuticals, MakScientific, Nano Precision Medical, NeonMind Biosciences, Novartis, Novo Nordisk, NuSirt Biopharma, Otsuka Pharmaceutical Factory Inc, Pfizer, Raziel Therapeutics, Reata Pharmaceuticals, Regeneron Pharmaceuticals, Reviva Pharmaceuticals, Rivus Pharmaceuticals, Shionogi, SHIONOGI & Co., Ltd., Sigrid Therapeutics, SJT Molecular Research SL, Structure Therapeutics, Sun Pharmaceutical Industries, Techfields Pharma, Terns Pharmaceuticals, Tonix Pharmaceuticals, UGISense AG, Versanis Bio, Viking Therapeutics, YSOPIA Bioscience, Yuhan, Zealand Pharma, and others.
- Promising Obesity Pipeline Therapies in the various stages of development include Semaglutide oral, Tirzepatide, Sibutramine/Topiramate, NNC0174-0833, Tesomet, SHR-20004, RZL-012, CT-868, PF-06882961, EMP-16, S-237648, LY3502970, ARD-101, NNC0165-1875, XW003, IBI362, TG103, Cotadutide, MBL949, PF-07081532, APHD-012, LY 3437943, HSG4112, Leucine/sildenafil, BI 456906, Pemvidutide, LY3841136, HM15136, GMA106, DD01, ZP8396, K833, NNC0480-0389, LB54640, CT-181, CB4211, CT-388, CIN-109, ERX-1000, DWP306001, SCO-094, S-309309, NO-13065, GDD3898, XW004, SCO-267, ASC41, AMG 133, LY3541105, AMG-786, HU6, BMS-963272, BI 1820237, Xla 1, Dapiglutide, PB-119, PB-718, AGTX-2004, ECC5004, Thermostem, XW017, HM 14320, TNX-2900, CF-102, XW 010, LR19020, LR19056, YH34160, NM-136, CIN 110, CIN 209, CIN 210, RSVI-301, PramExe, BC GlueExe, BC Pram GlueExe, AGEX BAT1, CLS-1, CRB-913, Tespria, AM 6545, LP-003, KT A522, JD5037, SJT4a, GL0034, TF0103, TERN-601, NEO-001, KTX 0200, ENT-03, Bimagrumab, VK2735, HUM-234, NPM 139, YHC2129, YHC2134, YHC1140, GSBR-2nd Gen, GSBR-3rd Gen, and others.
- December 2023: Rhythm Pharmaceuticals Inc. announced a study of Phase 3 clinical trials for Setmelanotide. The goal of this trial is to learn how well Setmelanotide works to improve weight reduction, hunger, and quality of life in patients 4 years of age and older with acquired Hypothalamic Obesity (HO). To determine how well setmelanotide works and how safe it is, patients with HO will take a daily injection of either setmelanotide or placebo and complete trial assessments for up to 60 weeks.
- December 2023: Pfizer announced a study of Phase 1 clinical trials for Rosuvastatin and Midazolam. A Phase 1 Study To Evaluate The Effect Of Two Steady State Doses of PF 06882961 On Rosuvastatin And Midazolam Pharmacokinetics In Otherwise Healthy Adult Participants With Obesity.
Request a sample and discover the recent advances in Obesity Treatment Drugs @ Obesity Pipeline Report
In the Obesity pipeline report, detailed description of the drug is given which includes the mechanism of action of the drug, clinical studies, NDA approvals (if any), and product development activities comprising the technology, Obesity collaborations, licensing, mergers and acquisition, funding, designations and other product related details.
Obesity Overview
Obesity is recognized as a chronic or non-communicable disease, a complex, multifactorial phenotype, affecting, along with overweight, primarily associated with excess adiposity or body fatness which can manifest metabolically and not just concerning the size of the body.
Find out more about Obesity Treatment Landscape @ Drugs for Obesity Treatment
Obesity Emerging Drugs Profile
- Semaglutide oral: Novo Nordisk
- Tirzepatide: Eli Lilly and Company
- Tesomet: Saniona
- RZL-012: Raziel Therapeutics
- CT-868: Carmot Therapeutics
- EMP16: Empros Pharma
- DD01: D&D Pharmatech
- ZP 8396: Zealand Pharma
- ERX-1000: ERX Pharmaceuticals
Obesity Pipeline Therapeutics Assessment
There are approx. 100+ key companies which are developing the therapies for Obesity. The Obesity companies which have their Obesity drug candidates in the most advanced stage, i.e. Phase III include, Novo Nordisk.
DelveInsight’s Obesity pipeline report covers around 130+ Obesity products under different phases of clinical development like
- Late stage products (Phase III)
- Mid-stage products (Phase II)
- Early-stage product (Phase I) along with the details of
- Pre-clinical and Discovery stage candidates
- Discontinued & Inactive candidates
Obesity pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Obesity Products have been categorized under various ROAs such as
- Oral
- Parenteral
- Intravenous
- Subcutaneous
- Topical.
Obesity Pipeline Products have been categorized under various Molecule types such as
- Monoclonal Antibody
- Peptides
- Polymer
- Small molecule
- Gene therapy
Learn more about the emerging Obesity Pipeline Therapies @ Obesity Clinical Trials Assessment
Scope of the Obesity Pipeline Report
- Coverage- Global
- Obesity Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
- Obesity Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
- Obesity Companies- 180 Life Sciences, 9 meters, Aardvark Therapeutics, Adocia, Agentix, AgeX Therapeutics, Altimmune, Amgen, Antag Therapeutics, Aphaia Pharma, Aptorum Group, AstraZeneca, Bukwang Pharmaceutical, Caliway Biopharmaceutics Co Ltd, Can Fite Biopharma, Carmot Therapeutics, Inc., Cellivery Therapeutics Inc, CinFina Pharma, Clayton Biotech, Click Therapeutics, Corbus Pharma, DiscoveryBiomed Inc, Dong-A ST, Dongkook Pharmaceuticals, Eccogene, Elevian, Eli Lilly and Company, Empros Pharma, Enterin Inc., EraCal Therapeutics AG, ERX Pharmaceuticals, ERX Pharmaceuticals, Eternygen GmbH, Eurofarma, Gannex Pharma, Glaceum, Gmax Biopharm, GPER G-1 Development Group, LLC, Gubra Therapeutics, Hanmi Pharmaceutical, Hanmi Pharmaceuticals, Innovent Biologics, Jenrin Discovery, Jiangsu Hansoh Pharmaceutical, Kallyope, Kintai Therapeutics, Kriya Therapeutics, LG Life Sciences, Lipidio Pharmaceuticals, MakScientific, Nano Precision Medical, NeonMind Biosciences, Novartis, Novo Nordisk, NuSirt Biopharma, Otsuka Pharmaceutical Factory Inc, Pfizer, Raziel Therapeutics, Reata Pharmaceuticals, Regeneron Pharmaceuticals, Reviva Pharmaceuticals, Rivus Pharmaceuticals, Shionogi, SHIONOGI & Co., Ltd., Sigrid Therapeutics, SJT Molecular Research SL, Structure Therapeutics, Sun Pharmaceutical Industries, Techfields Pharma, Terns Pharmaceuticals, Tonix Pharmaceuticals, UGISense AG, Versanis Bio, Viking Therapeutics, YSOPIA Bioscience, Yuhan, Zealand Pharma, and others.
- Obesity Pipeline Therapies- Semaglutide oral, Tirzepatide, Sibutramine/Topiramate, NNC0174-0833, Tesomet, SHR-20004, RZL-012, CT-868, PF-06882961, EMP-16, S-237648, LY3502970, ARD-101, NNC0165-1875, XW003, IBI362, TG103, Cotadutide, MBL949, PF-07081532, APHD-012, LY 3437943, HSG4112, Leucine/sildenafil, BI 456906, Pemvidutide, LY3841136, HM15136, GMA106, DD01, ZP8396, K833, NNC0480-0389, LB54640, CT-181, CB4211, CT-388, CIN-109, ERX-1000, DWP306001, SCO-094, S-309309, NO-13065, GDD3898, XW004, SCO-267, ASC41, AMG 133, LY3541105, AMG-786, HU6, BMS-963272, BI 1820237, Xla 1, Dapiglutide, PB-119, PB-718, AGTX-2004, ECC5004, Thermostem, XW017, HM 14320, TNX-2900, CF-102, XW 010, LR19020, LR19056, YH34160, NM-136, CIN 110, CIN 209, CIN 210, RSVI-301, PramExe, BC GlueExe, BC Pram GlueExe, AGEX BAT1, CLS-1, CRB-913, Tespria, AM 6545, LP-003, KT A522, JD5037, SJT4a, GL0034, TF0103, TERN-601, NEO-001, KTX 0200, ENT-03, Bimagrumab, VK2735, HUM-234, NPM 139, YHC2129, YHC2134, YHC1140, GSBR-2nd Gen, GSBR-3rd Gen, and others.
Dive deep into rich insights for new drugs for Obesity treatment, Visit @ Obesity Market Drivers and Barriers, and Future Perspective
Table of Content
- Introduction
- Executive Summary
- Obesity Overview
- Obesity Pipeline Therapeutics
- Obesity Therapeutic Assessment
- Late Stage Products (Phase III)
- Oral Semaglutide: Novo Nordisk
- Drug profiles in the detailed report…..
- Mid Stage Products (Phase II)
- RZL-012: Raziel Therapeutics
- Drug profiles in the detailed report…..
- Early Stage Products (Phase I)
- DD01: D&D Pharmatech
- Drug profiles in the detailed report…..
- Inactive Obesity Products
- Obesity Key Companies
- Obesity Key Products
- Obesity Unmet Needs
- Obesity Market Drivers
- Obesity Market Barriers
- Obesity Future Perspectives and Conclusion
- Obesity Analyst Views
- Obesity Key Companies
- Appendix
For further information on the Obesity pipeline therapeutics, reach out @ Obesity Unmet Needs and Analyst Views
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email: Send Email
Phone: 9193216187
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: NV
Country: United States
Website: https://www.delveinsight.com/report-store/familial-lipoprotein-lipase-deficiency-market